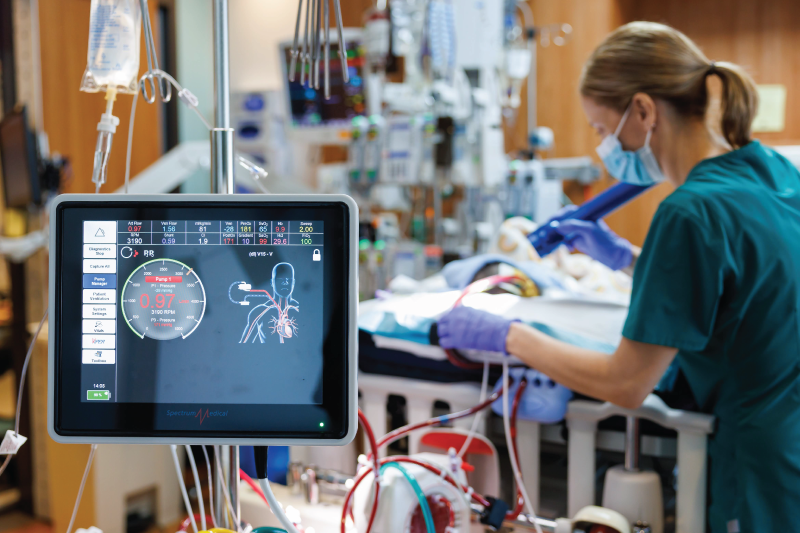
The pulmonary vein and artery system (Stock image)
Pulmonary vein stenosis (PVS) is a rare yet serious cardiovascular condition marked by progressive narrowing of the veins that return oxygenated blood from the lungs to the heart. Until recently, it was usually considered a death sentence. “There’s no two ways about it,” said Children’s of Alabama pediatric cardiologist Matt Clark, M.D. “It only gets worse once it starts.”
“When diagnosed, we would tell families there was nothing more to offer and to transition to end-of-life type care,” added pediatric interventional cardiologist Stephen Clark, M.D. “Until recently, we didn’t have medical therapies that could treat this. Surgical intervention was generally unsuccessful and sometimes accelerated the progression of the disease.”
Indeed, PVS has historically had grim outcomes, with mortality rates as high as 60% or more. Right ventricular pressure elevation—a marker of pulmonary hypertension (PH)—is strongly associated with mortality.
Now, there is hope.
“I think the biggest thing to come along was more data from studies and centers showing if you change the mindset and consider it as a possibly treatable condition, you change outcomes,” Matt Clark said.
That shift in mindset has driven a transformation in how PVS is diagnosed, monitored and treated. Today, children who would not have survived infancy are living into early childhood—and in some cases beyond—because of earlier recognition, aggressive catheter-based interventions and multidisciplinary care.
Earlier Detection, Closer Surveillance
One of the most important changes in PVS care has been earlier diagnosis, Matt Clark said, by recognizing high-risk populations: neonates with chronic lung disease; prolonged ventilation; those with congenital heart disease; and/or those with pulmonary hypertension (PH). The earlier the diagnosis, the more likely interventions are to be effective.
This surveillance-driven approach means clinicians are often identifying milder disease earlier—with regular echocardiograms, CT scans and diagnostic catheterization—and intervening at the first signs of progression.
PVS often exists in the setting of comorbid conditions such as PH, congenital heart disease or complications of prematurity, requiring close coordination between specialists. Thus, the pulmonary vein stenosis team in the Children’s of Alabama Heart Center has brought together a small group of pediatric cardiologists, allowing for shared memory and standardized decision-making.
“We don’t have to spend time reinventing the wheel,” Matt Clark said. “We can collectively remember all our patients” and which approaches approach worked best.
Catheter-Based Intervention as a Strategy, Not a Last Resort
Treatment for PVS has also evolved. Intervention includes balloon angioplasty to widen narrowed vessels or stent placement to hold them open. The stents are coated with sirolimus, a drug designed to prevent restenosis. However, rather than viewing catheter-based intervention to unblock the vessel as a one-time attempt to rescue a sick child, it is now viewed as a repeated, planned strategy.
“Every time you do an intervention,” Matt Clark said, “you’re signing that kid up for another cath at some interval.” That’s because the baby’s heart and lungs keep growing, requiring additional interventions. Multicenter analyses show that repeat catheterizations are associated with improved survival, reflecting a shift away from passive observation to active disease control.
In some children, repeated dilations and stent expansions over time allow pulmonary veins to reach near-adult size within the first few years of life.
Medical Therapy to Slow Disease Progression
Procedural care is now complemented by medical therapy aimed at the underlying biology of PVS.
Because the disease involves aggressive cellular proliferation within the vessel wall, antiproliferative medications such as sirolimus have become an important adjunct to intervention.
Observational studies suggest that children receiving systemic sirolimus have significantly improved survival compared with historical controls. In one widely cited cohort, four-year survival reached 100% in treated patients compared with approximately 45% in those not receiving therapy.
Sirolimus has also been associated with slower in-stent restenosis and longer intervals between interventions. While randomized trials are still lacking, these findings have helped legitimize systemic therapy as part of standard care at experienced centers.
“We’re probably doing more sirolimus-type therapies now than we used to,” Matt Clark said.
Survival Looks Different Now
Even with advances, PVS remains unpredictable. Not every child can be saved. But survival is no longer measured only in weeks or months.
Matt Clark described children who once would have died in the hospital now going home, growing and returning for planned interventions.
“One of the earlier ones was pretty impactful,” he said, recalling a child who lived long enough to spend two Christmases at home. “That was enough to make us keep going and keep pushing.”
“We know we’re helping,” he said. “So we’re more likely to intervene, to follow closely and to keep trying.”