Children’s neonatologists work with surgeons on many types of complex cases. (File photo)
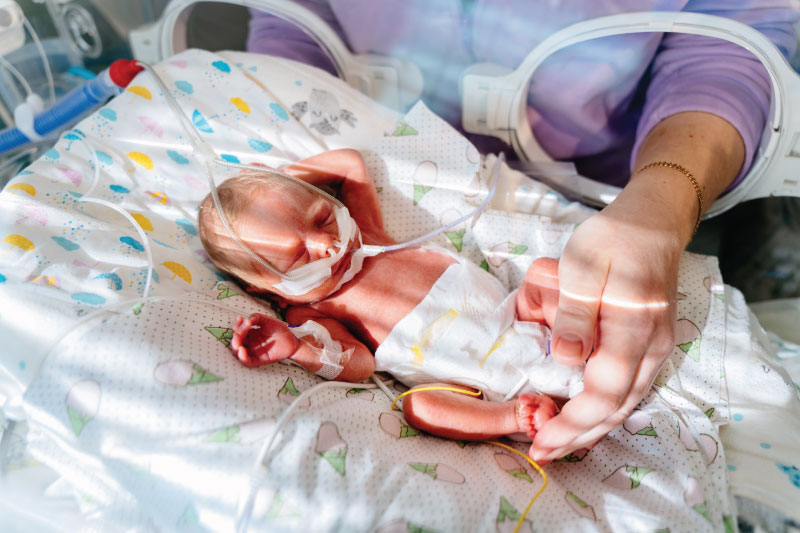
In the Level IV Neonatal Intensive Care Unit (NICU), Children’s of Alabama cares for some of the sickest neonates in Alabama and the surrounding region. The unit, which expanded in 2025, frequently receives referrals from other hospitals for surgical and subspecialty care. Often, the babies cared for on the unit have conditions that require complex surgeries. In these cases, neonatologists work with surgeons and other specialists to manage the patient’s care. Jaw distraction is one example of a procedure they may manage.
Jaw Distraction Surgery and the Role of Neonatology
For some infants, a small or recessed jaw creates serious problems with breathing and feeding. Jaw distraction surgery—also called mandibular distraction osteogenesis—is an option that may dramatically improve outcomes for these patients. While the surgery itself is performed by craniofacial surgeons, the care before and after is deeply collaborative. Neonatologists play a central role in guiding babies and families through every step of the process.
Physicians typically consider jaw distraction surgery for young infants, often within their first month or two of life. Babies with the condition often work harder to breathe normally. They also may have difficulty feeding by mouth because the small jaw narrows the airway. If growth alone doesn’t lead to improvement, then a multidisciplinary team, including neonatologists, craniofacial surgeons, and often ear, nose, and throat (ENT) surgeons, evaluates whether jaw distraction could improve their symptoms and quality of life.
Surgeons focus on the technical aspects of lengthening the jaw, but before and after the operating room, neonatologists manage the airway, ventilation, nutrition and pain control. After the procedure, the baby returns to the NICU sedated and temporarily paralyzed. This protects the infant’s airway and allows the surgical site to heal while the jaw is gradually lengthened over several days. During this time, infants require ventilator support, careful airway management, pain control and nutritional support.
“This isn’t a procedure we recommend lightly,” Children’s of Alabama neonatologist Hannah Hightower, M.D., explained. “Protecting the airway is our number one concern for infants who undergo this surgery.” Because surgeons place hardware in the jaw, any movement could risk complications. This also means post-op babies cannot be held. This period can be especially hard on new parents who may have already begun holding and feeding their child.
As recovery progresses, collaboration becomes increasingly important. Neonatologists and surgeons work together to determine when it is safe to reduce sedation, remove the breathing tube and transition the baby off respiratory support. “Then, we begin working on the infant’s oral skills, because our ultimate goal is for the family to be able to feed their baby at home,” Hightower said. Many of these infants require tube feeding at first, but most eventually learn to feed by mouth.
Even in ideal cases, infants typically remain hospitalized for several weeks as they recover, learn to eat and prepare for discharge. Parents play a significant role as the baby’s recovery progresses, becoming actively involved in feeding and comforting their child. Success after jaw distraction is measured in practical, meaningful ways, such as improved breathing without respiratory support and the ability to take feeds by bottle.
Though the procedure can seem daunting, Hightower emphasizes its impact. Jaw distraction surgery can transform the course of an infant’s life—turning a situation marked by breathing struggles and feeding challenges into one where a baby can go home, breathe comfortably and be fed by their family. “It’s remarkable how a child who is working to breathe and unable to eat may improve and gain these skills post-surgery,” Hightower said. “Being able to send a child home doing those normal baby things is a big deal for the parents.”