Research shows that the gut composition of fungi in the second week of life predicts the later development of BPD.
By Jeff Hansen (UAB)
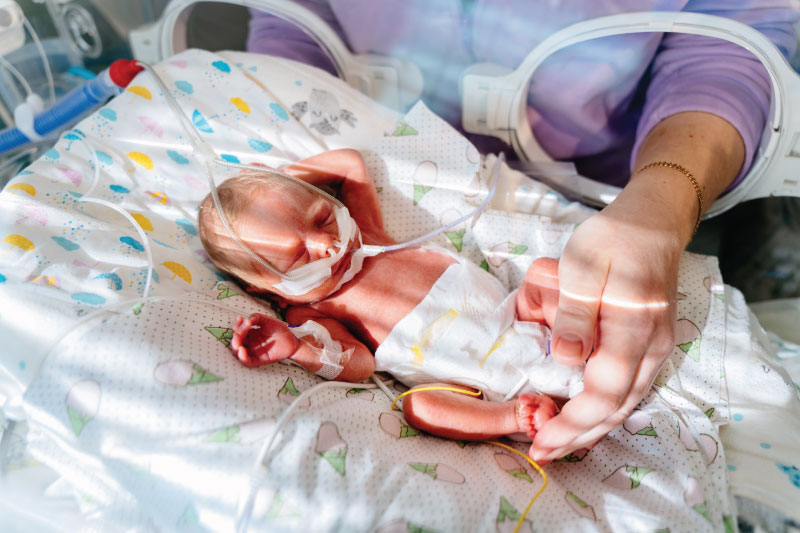
Extremely preterm newborns who weigh less than 3.3 pounds have immature lungs that often require high levels of ventilation oxygen in the hospital. This contributes to the chronic lung disease bronchopulmonary dysplasia, or BPD, the most common cause of death for these tiny infants. BPD exacts a devastating toll on the immature lung.
In one of the most extensive studies of the microorganisms in the intestines of very preterm infants, University of Alabama at Birmingham (UAB) and University of Tennessee Health Science Center researchers show that the gut composition of fungi in the second week of life predicts the later development of BPD, weeks to months before diagnosis of that disease. They analyzed gut fungi in the first true non-meconium stool produced before two weeks of life and found that the fungal intestinal microbiome—known as the mycobiome—of infants who later developed BPD differed in community diversity, composition and interconnectivity from the infants who never got BPD, as measured by the most up-to-date bioinformatic techniques. The researchers did not find significant differences in the bacterial microbiome in those first true stools.
To show causality, researchers transferred samples of the first true stool that predicts BPD or the first true stool of newborns who did not get BPD into female mice to give them a pseudo-humanized gut microflora. In a mouse model of BPD, newborn pups from those BPD dams showed an increased in the severity of lung injury compared with newborn pups from the no-BPD dams. In loss-of-function experiments, when the female mice with the BPD-predictive stool transplant were treated with an antifungal agent before birth, that inhibition of perinatal fungal colonization reduced lung injury in the newborn pups. In contrast, a gain-of-function experiment, where the perinatal fungal colonization of dams was augmented with a species of Candida fungus common in mice, amplified BPD severity in the newborn pups.

“These findings demonstrate that features of the initial intestinal fungal microbiome are associated with the later development of BPD in premature neonates and exert a microbiome-driven effect that is transferable and modifiable in mouse models,” said Children’s of Alabama neonatologist Kent Willis, M.D., who’s also an assistant professor in the UAB Department of Pediatrics Division of Neonatology. “This suggests causality, and it suggests that the gut fungi may represent a therapeutic target in newborn lung disease.”
Willis and Ajay J. Talati, M.D., University of Tennessee Health Science Center, Memphis, Tennessee, co-led the study, published in Microbiome.
“Collectively, our analyses demonstrate that the composition of the intestinal mycobiome of infants who did not develop BPD was more uniform,” Willis said. “In contrast, those who eventually developed BPD had more disparate mycobiomes. This suggests that a particular pattern of mycobiome development may be necessary to impart resistance to the development of BPD, and failure to do so in various ways is associated with disease development.”
The first defecations of newborn infants are meconium, composed from materials ingested while in the uterus. The first true stools in the second week, the ones analyzed by Willis and Talati, are digested milk. It is known, Willis and Talati say, that fungi in adults are vital members of the human microbiota; but compared to bacteria, their non-pathological and non-parasitic functions are still poorly understood, especially in newborns.
This prospective observational cohort study included newborn stool samples collected over six years from 2017-2020 in Memphis and 2021-2022 in Birmingham. The 64 very-preterm infants in the study who did not develop BPD had an average birthweight of 2.5 pounds, and the 38 very-preterm infants who did develop BPD had an average birthweight of 1.6 pounds. Only one of the 64 no-BPD infants in the study died, while six of the 38 BPD infants died.